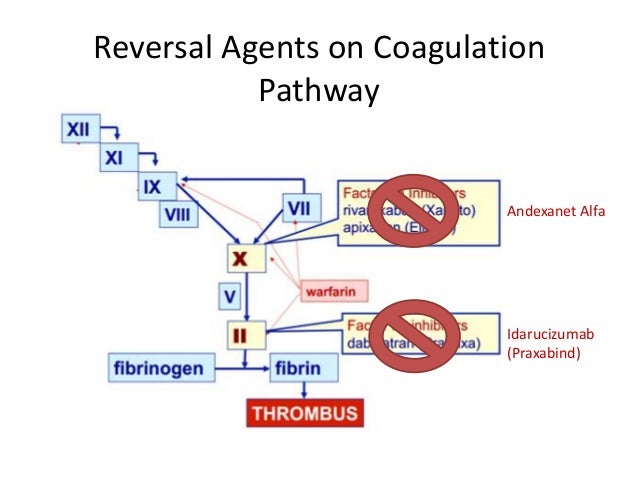 
Furthermore, the data provided, including some data on reduced bleeding, were considered promising. However, the Agency also noted the unmet medical need to stop life-threatening or uncontrolled bleeding caused by factor Xa inhibitors. Patients treated with Ondexxya, especially those aged over 75 years, may be at a higher risk of thromboembolism. The European Medicines Agency noted that it has not been established that antifactor Xa activity can be used as a reliable measure for reduced bleeding and that there was not enough evidence on the use of Ondexxya to reverse the effects of edoxaban, another factor Xa inhibitor. On the basis of studies in healthy volunteers and preliminary data from studies in patients, Ondexxya was found effective for reducing antifactor Xa activity in those taking anticoagulants that block factor Xa (factor Xa inhibitors). What are the risks associated with Ondexxya? After treatment with Ondexxya, antifactor Xa activity was reduced on average by 90% in 83 patients taking apixaban and by 78% in 70 patients taking rivaroxaban. Involving people who had taken rivaroxaban, the full dose of Ondexxya reduced antifactor Xa activity by 97% on average in 26 people, compared with 45% in 13 people receiving placebo.Ī further ongoing study involved patients taking a factor Xa inhibitor anticoagulant who had major bleeding. In the first study, involving people who had taken apixaban, the full dose of Ondexxya reduced antifactor Xa activity by 92% on average in 23 people compared with 33% in 8 people receiving placebo (a dummy treatment). The main measure of effectiveness was the reduction in 'antifactor Xa activity' (a measure of how well natural factor Xa is blocked by an anticoagulant). Two main studies have found Ondexxya effective in healthy volunteers who took either apixaban or rivaroxaban, two anticoagulants that block the effect of factor Xa.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
